QUICK NAV:
Causes of Corrosion
Corrosion is a natural process. The primary driving force of corrosion is based upon the transformation of iron from its natural state to steel. The refining of iron ore into steel requires the addition of energy. Steel is essentially an unstable state of iron and corrosion is the process of iron returning to its natural state. The energy used in the refining process is the driving force of corrosion.
Corrosion cells are established on underground pipelines for a variety of causes. A primary cause of corrosion is due to an effect known as galvanic corrosion. All metals have different natural electrical potentials. Where two metals with different potentials are connected to each other in a common environment, current will flow causing corrosion to occur. The coupling of steel to a different metal, such as copper, will cause a corrosion circuit to be established. Direct coupling of copper to steel will cause the steel to corrode much faster than normal. Another form of this is the coupling of rusty pipe to new, clean steel. The natural difference in potential causes the new steel to corrode more rapidly than the old steel. Other causes of pipeline corrosion cells include the effect of different soil types, oxygen availability, stray current interference and microbiological growth.
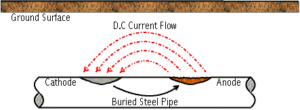
Two other unique causes (and sometimes related) are stress and hydrogen.
- Stress Corrosion occurs when even a very small pit forms in a metal under stress. The concentrated stress either deepens and extends the pit, or cracks the protective film which tends to form. Under continued exposure to the corrosive medium and stress, the crack extends by alternate corrosion and stress failure.
- Hydrogen Embrittlement and hydrogen attack results when atomic hydrogen penetrates into the grain boundaries of steel producing microcracks, blistering and loss of ductility. The atomic hydrogen combines into molecules and results in blistering and laminations.